HAEM5:B-lymphoblastic leukaemia/lymphoma with iAMP21: Difference between revisions
| [checked revision] | [checked revision] |
| Line 40: | Line 40: | ||
==Gene Rearrangements== | ==Gene Rearrangements== | ||
Some rearrangements have been seen as secondary abnormalities. | Some rearrangements have been seen as secondary abnormalities. | ||
{| class="wikitable sortable" | {| class="wikitable sortable" | ||
|- | |- | ||
! | !Driver Gene!!Fusion(s) and Common Partner Genes!!Molecular Pathogenesis!!Typical Chromosomal Alteration(s) | ||
! | !Prevalence -Common >20%, Recurrent 5-20% or Rare <5% (Disease) | ||
!Prognostic Significance | !Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | ||
! | !Established Clinical Significance Per Guidelines - Yes or No (Source) | ||
!Notes | !Clinical Relevance Details/Other Notes | ||
|- | |- | ||
| | |''CRLF2'' | ||
|''P2RY8::CRLF2'' | |||
| | | | ||
|del(X)(p22.33p22.33)/del(Y)(p11.32p11.32) | |||
| | | | ||
| | | | ||
|Because of the unique nature of the iAMP21 abnormality, cases that present with additional genomic lesions that may suggest another category, such as a CRLF2 rearrangement, should still be classified as B-ALL with iAMP21. | | | ||
| rowspan="3" |Because of the unique nature of the iAMP21 abnormality, cases that present with additional genomic lesions that may suggest another category, such as a ''CRLF2'' rearrangement, should still be classified as B-ALL with iAMP21. | |||
|- | |- | ||
|Chimeric transcription factor/fusion gene | |||
|''ETV6::RUNX1'' | |||
|The driver gene is a result of the fusion of 5'<nowiki/>''ETV6'' and 3'''RUNX1,'' genes that regulate hematopoiesis, which creates a chimeric transcription factor. | |||
|t(12;21)(p13.2;q22.1) | |t(12;21)(p13.2;q22.1) | ||
| | | | ||
| | | | ||
| | | | ||
|- | |||
|''ABL'' | |||
|''BCR::ABL1'' | |||
|The pathogenic derivative is the der(22) resulting in fusion of 5’ ''BCR'' and 3’''ABL1''. | |||
|t(9;22)(q34;q11.2) | |||
| | |||
| | |||
| | |||
|} | |||
==Individual Region Genomic Gain/Loss/LOH== | |||
{| class="wikitable sortable" | |||
|- | |||
!Chr #!!Gain, Loss, Amp, LOH!!Minimal Region Cytoband and/or Genomic Coordinates [Genome Build; Size]!!Relevant Gene(s) | |||
!Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | |||
!Established Clinical Significance Per Guidelines - Yes or No (Source) | |||
!Clinical Relevance Details/Other Notes | |||
|- | |||
|<span class="blue-text">EXAMPLE:</span>7 | |||
|<span class="blue-text">EXAMPLE:</span> Loss | |||
|<span class="blue-text">EXAMPLE:</span>chr7 | |||
|<span class="blue-text">EXAMPLE:</span>Unknown | |||
|<span class="blue-text">EXAMPLE:</span> D, P | |||
|<span class="blue-text">EXAMPLE:</span> No | |||
|<span class="blue-text">EXAMPLE:</span>Presence of monosomy 7 (or 7q deletion) is sufficient for a diagnosis of AML with MDS-related changes when there is ≥20% blasts and no prior therapy (add reference). Monosomy 7/7q deletion is associated with a poor prognosis in AML (add references). | |||
|- | |||
|<span class="blue-text">EXAMPLE:</span>8 | |||
|<span class="blue-text">EXAMPLE:</span> Gain | |||
|<span class="blue-text">EXAMPLE:</span>chr8 | |||
|<span class="blue-text">EXAMPLE:</span>Unknown | |||
|<span class="blue-text">EXAMPLE:</span> D, P | |||
| | | | ||
|<span class="blue-text">EXAMPLE:</span>Common recurrent secondary finding for t(8;21) (add references). | |||
|- | |||
|<span class="blue-text">EXAMPLE:</span>17 | |||
|<span class="blue-text">EXAMPLE:</span> Amp | |||
|<span class="blue-text">EXAMPLE:</span>17q12; chr17:39,700,064-39,728,658 [hg38; 28.6 kb] | |||
|<span class="blue-text">EXAMPLE:</span>''ERBB2'' | |||
|<span class="blue-text">EXAMPLE:</span> D, P, T | |||
| | | | ||
|<span class="blue-text">EXAMPLE:</span>Amplification of ''ERBB2'' is associated with HER2 overexpression in HER2 positive breast cancer (add references). Add criteria for how amplification is defined. | |||
|- | |- | ||
| | | | ||
| | | | ||
| Line 75: | Line 111: | ||
| | | | ||
| | | | ||
| | | | ||
| | |||
|}Cytogenetic morphology of the abnormal chromosome 21 can vary markedly between patients.<ref name=":3">{{Cite journal|last=Harewood|first=L.|last2=Robinson|first2=H.|last3=Harris|first3=R.|last4=Al-Obaidi|first4=M. Jabbar|last5=Jalali|first5=G. R.|last6=Martineau|first6=M.|last7=Moorman|first7=A. V.|last8=Sumption|first8=N.|last9=Richards|first9=S.|date=2003-03|title=Amplification of AML1 on a duplicated chromosome 21 in acute lymphoblastic leukemia: a study of 20 cases|url=https://pubmed.ncbi.nlm.nih.gov/12646943|journal=Leukemia|volume=17|issue=3|pages=547–553|doi=10.1038/sj.leu.2402849|issn=0887-6924|pmid=12646943}}</ref> In ~80% of iAMP21 B-ALL cases, recurrent secondary abnormalities, both chromosomal and molecular, have been documented. Deletions involving particular genes such as; ''IKZF1, CDKN2A/B, PAX5, SH2B3, ETV6'' and ''RB1'' have also been observed.[[File:IAMP21 met.jpg|center|thumb|875x875px|iAMP21 in a ring formation; Courtesy of Fullerton Genetics Lab]] | |||
Cytogenetic morphology of the abnormal chromosome 21 can vary markedly between patients.<ref name=":3">{{Cite journal|last=Harewood|first=L.|last2=Robinson|first2=H.|last3=Harris|first3=R.|last4=Al-Obaidi|first4=M. Jabbar|last5=Jalali|first5=G. R.|last6=Martineau|first6=M.|last7=Moorman|first7=A. V.|last8=Sumption|first8=N.|last9=Richards|first9=S.|date=2003-03|title=Amplification of AML1 on a duplicated chromosome 21 in acute lymphoblastic leukemia: a study of 20 cases|url=https://pubmed.ncbi.nlm.nih.gov/12646943|journal=Leukemia|volume=17|issue=3|pages=547–553|doi=10.1038/sj.leu.2402849|issn=0887-6924|pmid=12646943}}</ref> | |||
In ~80% of iAMP21 B-ALL cases, recurrent secondary abnormalities, both chromosomal and molecular, have been documented. Deletions involving particular genes such as; ''IKZF1, CDKN2A/B, PAX5, SH2B3, ETV6'' and ''RB1'' have also been observed. | |||
==Characteristic Chromosomal or Other Global Mutational Patterns== | ==Characteristic Chromosomal or Other Global Mutational Patterns== | ||
| Line 91: | Line 119: | ||
iAMP21 cases have a characteristic pattern that is both complex and variable. This pattern comprises multiple regions of gain, amplification and deletion. Interestingly, ''RUNX1'' amplification is not always intrachromosomal.<ref>{{Cite journal|last=Arber|first=Daniel A.|date=04 2019|title=The 2016 WHO classification of acute myeloid leukemia: What the practicing clinician needs to know|url=https://pubmed.ncbi.nlm.nih.gov/30926096|journal=Seminars in Hematology|volume=56|issue=2|pages=90–95|doi=10.1053/j.seminhematol.2018.08.002|issn=1532-8686|pmid=30926096}}</ref><ref>{{Cite journal|last=Johnson|first=Ryan C.|last2=Weinberg|first2=Olga K.|last3=Cascio|first3=Michael J.|last4=Dahl|first4=Gary V.|last5=Mitton|first5=Bryan A.|last6=Silverman|first6=Lewis B.|last7=Cherry|first7=Athena M.|last8=Arber|first8=Daniel A.|last9=Ohgami|first9=Robert S.|date=2015-07|title=Cytogenetic Variation of B-Lymphoblastic Leukemia With Intrachromosomal Amplification of Chromosome 21 (iAMP21): A Multi-Institutional Series Review|url=https://pubmed.ncbi.nlm.nih.gov/26071468|journal=American Journal of Clinical Pathology|volume=144|issue=1|pages=103–112|doi=10.1309/AJCPLUYF11HQBYRB|issn=1943-7722|pmid=26071468}}</ref> The formation of iAMP21 is considered to be due to breakage-fusion-bridge cycles followed by chromothripsis and other complex structural rearrangements of chromosome 21. Studies, molecular and cytogenetic, have elucidated a common 5.1 Mb region that includes the ''RUNX1'' gene. However, even though ''RUNX1'' is included in the amplified region, there has not yet been any conclusive evidence that ''RUNX1'' is critical in the pathogenesis of disease given that it is not overexpressed in some individuals with this abnormality.<ref name=":0">{{Cite journal|last=Akkari|first=Yassmine M. N.|last2=Bruyere|first2=Helene|last3=Hagelstrom|first3=R. Tanner|last4=Kanagal-Shamanna|first4=Rashmi|last5=Liu|first5=Jie|last6=Luo|first6=Minjie|last7=Mikhail|first7=Fady M.|last8=Pitel|first8=Beth A.|last9=Raca|first9=Gordana|date=2020-05|title=Evidence-based review of genomic aberrations in B-lymphoblastic leukemia/lymphoma: Report from the cancer genomics consortium working group for lymphoblastic leukemia|url=https://pubmed.ncbi.nlm.nih.gov/32302940|journal=Cancer Genetics|volume=243|pages=52–72|doi=10.1016/j.cancergen.2020.03.001|issn=2210-7762|pmid=32302940}}</ref><ref>{{Cite journal|last=Rand|first=Vikki|last2=Parker|first2=Helen|last3=Russell|first3=Lisa J.|last4=Schwab|first4=Claire|last5=Ensor|first5=Hannah|last6=Irving|first6=Julie|last7=Jones|first7=Lisa|last8=Masic|first8=Dino|last9=Minto|first9=Lynne|date=2011-06-23|title=Genomic characterization implicates iAMP21 as a likely primary genetic event in childhood B-cell precursor acute lymphoblastic leukemia|url=https://pubmed.ncbi.nlm.nih.gov/21527530|journal=Blood|volume=117|issue=25|pages=6848–6855|doi=10.1182/blood-2011-01-329961|issn=1528-0020|pmid=21527530}}</ref><ref>{{Cite journal|last=Hunger|first=Stephen P.|last2=Lu|first2=Xiaomin|last3=Devidas|first3=Meenakshi|last4=Camitta|first4=Bruce M.|last5=Gaynon|first5=Paul S.|last6=Winick|first6=Naomi J.|last7=Reaman|first7=Gregory H.|last8=Carroll|first8=William L.|date=2012-05-10|title=Improved survival for children and adolescents with acute lymphoblastic leukemia between 1990 and 2005: a report from the children's oncology group|url=https://pubmed.ncbi.nlm.nih.gov/22412151|journal=Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology|volume=30|issue=14|pages=1663–1669|doi=10.1200/JCO.2011.37.8018|issn=1527-7755|pmc=3383113|pmid=22412151}}</ref> | iAMP21 cases have a characteristic pattern that is both complex and variable. This pattern comprises multiple regions of gain, amplification and deletion. Interestingly, ''RUNX1'' amplification is not always intrachromosomal.<ref>{{Cite journal|last=Arber|first=Daniel A.|date=04 2019|title=The 2016 WHO classification of acute myeloid leukemia: What the practicing clinician needs to know|url=https://pubmed.ncbi.nlm.nih.gov/30926096|journal=Seminars in Hematology|volume=56|issue=2|pages=90–95|doi=10.1053/j.seminhematol.2018.08.002|issn=1532-8686|pmid=30926096}}</ref><ref>{{Cite journal|last=Johnson|first=Ryan C.|last2=Weinberg|first2=Olga K.|last3=Cascio|first3=Michael J.|last4=Dahl|first4=Gary V.|last5=Mitton|first5=Bryan A.|last6=Silverman|first6=Lewis B.|last7=Cherry|first7=Athena M.|last8=Arber|first8=Daniel A.|last9=Ohgami|first9=Robert S.|date=2015-07|title=Cytogenetic Variation of B-Lymphoblastic Leukemia With Intrachromosomal Amplification of Chromosome 21 (iAMP21): A Multi-Institutional Series Review|url=https://pubmed.ncbi.nlm.nih.gov/26071468|journal=American Journal of Clinical Pathology|volume=144|issue=1|pages=103–112|doi=10.1309/AJCPLUYF11HQBYRB|issn=1943-7722|pmid=26071468}}</ref> The formation of iAMP21 is considered to be due to breakage-fusion-bridge cycles followed by chromothripsis and other complex structural rearrangements of chromosome 21. Studies, molecular and cytogenetic, have elucidated a common 5.1 Mb region that includes the ''RUNX1'' gene. However, even though ''RUNX1'' is included in the amplified region, there has not yet been any conclusive evidence that ''RUNX1'' is critical in the pathogenesis of disease given that it is not overexpressed in some individuals with this abnormality.<ref name=":0">{{Cite journal|last=Akkari|first=Yassmine M. N.|last2=Bruyere|first2=Helene|last3=Hagelstrom|first3=R. Tanner|last4=Kanagal-Shamanna|first4=Rashmi|last5=Liu|first5=Jie|last6=Luo|first6=Minjie|last7=Mikhail|first7=Fady M.|last8=Pitel|first8=Beth A.|last9=Raca|first9=Gordana|date=2020-05|title=Evidence-based review of genomic aberrations in B-lymphoblastic leukemia/lymphoma: Report from the cancer genomics consortium working group for lymphoblastic leukemia|url=https://pubmed.ncbi.nlm.nih.gov/32302940|journal=Cancer Genetics|volume=243|pages=52–72|doi=10.1016/j.cancergen.2020.03.001|issn=2210-7762|pmid=32302940}}</ref><ref>{{Cite journal|last=Rand|first=Vikki|last2=Parker|first2=Helen|last3=Russell|first3=Lisa J.|last4=Schwab|first4=Claire|last5=Ensor|first5=Hannah|last6=Irving|first6=Julie|last7=Jones|first7=Lisa|last8=Masic|first8=Dino|last9=Minto|first9=Lynne|date=2011-06-23|title=Genomic characterization implicates iAMP21 as a likely primary genetic event in childhood B-cell precursor acute lymphoblastic leukemia|url=https://pubmed.ncbi.nlm.nih.gov/21527530|journal=Blood|volume=117|issue=25|pages=6848–6855|doi=10.1182/blood-2011-01-329961|issn=1528-0020|pmid=21527530}}</ref><ref>{{Cite journal|last=Hunger|first=Stephen P.|last2=Lu|first2=Xiaomin|last3=Devidas|first3=Meenakshi|last4=Camitta|first4=Bruce M.|last5=Gaynon|first5=Paul S.|last6=Winick|first6=Naomi J.|last7=Reaman|first7=Gregory H.|last8=Carroll|first8=William L.|date=2012-05-10|title=Improved survival for children and adolescents with acute lymphoblastic leukemia between 1990 and 2005: a report from the children's oncology group|url=https://pubmed.ncbi.nlm.nih.gov/22412151|journal=Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology|volume=30|issue=14|pages=1663–1669|doi=10.1200/JCO.2011.37.8018|issn=1527-7755|pmc=3383113|pmid=22412151}}</ref> | ||
{| class="wikitable sortable" | {| class="wikitable sortable" | ||
|- | |- | ||
!Chromosomal Pattern | !Chromosomal Pattern | ||
! | !Molecular Pathogenesis | ||
!Prognostic Significance | !Prevalence - | ||
! | Common >20%, Recurrent 5-20% or Rare <5% (Disease) | ||
!Notes | !Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | ||
!Established Clinical Significance Per Guidelines - Yes or No (Source) | |||
!Clinical Relevance Details/Other Notes | |||
|- | |||
|<span class="blue-text">EXAMPLE:</span>Co-deletion of 1p and 18q | |||
|<span class="blue-text">EXAMPLE:</span> See chromosomal rearrangements table as this pattern is due to an unbalanced derivative translocation associated with oligodendroglioma (add reference). | |||
|<span class="blue-text">EXAMPLE:</span> Common (Oligodendroglioma) | |||
|<span class="blue-text">EXAMPLE:</span> D, P | |||
| | |||
| | |||
|- | |||
|<span class="blue-text">EXAMPLE:</span>Microsatellite instability - hypermutated | |||
| | |||
|<span class="blue-text">EXAMPLE:</span> Common (Endometrial carcinoma) | |||
|<span class="blue-text">EXAMPLE:</span> P, T | |||
| | |||
| | |||
|- | |- | ||
|Amplification of RUNX1 | |Amplification of ''RUNX1'' | ||
| | | | ||
| | | | ||
|D | |||
| | | | ||
|See CMA image displaying the amplification of RUNX1. This is part of the critical region consistently amplified: chr21:32.8-37.9 Mb (hg19). | |See CMA image displaying the amplification of RUNX1. This is part of the critical region consistently amplified: chr21:32.8-37.9 Mb (hg19). | ||
| Line 111: | Line 155: | ||
| | | | ||
| | | | ||
|} | | | ||
[[File:IAMP21 CMA .png|center|thumb|1028x1028px|Characteristic iAMP21 CMA; Courtesy of Fullerton Genetics Lab]] | |- | ||
|Gain of X chromosome | |||
| | |||
| | |||
| | |||
| | |||
| | |||
|- | |||
|Abnormalities of chromosomes 7 and 11 | |||
| | |||
| | |||
| | |||
| | |||
| | |||
|- | |||
|Deletions of ''RB1'' and ''ETV6'' | |||
| | |||
| | |||
| | |||
| | |||
| | |||
|}[[File:IAMP21 CMA .png|center|thumb|1028x1028px|Characteristic iAMP21 CMA; Courtesy of Fullerton Genetics Lab]] | |||
<br /> | <br /> | ||
| Line 119: | Line 184: | ||
In a 2016 paper, it was shown that in the iAMP21-ALL exome, the mutations were more commonly transitions (for example: C>T) than transversions or indels.<ref name=":0" /><ref name=":2">{{Cite journal|last=Ryan|first=S. L.|last2=Matheson|first2=E.|last3=Grossmann|first3=V.|last4=Sinclair|first4=P.|last5=Bashton|first5=M.|last6=Schwab|first6=C.|last7=Towers|first7=W.|last8=Partington|first8=M.|last9=Elliott|first9=A.|date=09 2016|title=The role of the RAS pathway in iAMP21-ALL|url=https://pubmed.ncbi.nlm.nih.gov/27168466|journal=Leukemia|volume=30|issue=9|pages=1824–1831|doi=10.1038/leu.2016.80|issn=1476-5551|pmc=5017527|pmid=27168466}}</ref> Frequently, mutations in the RAS signaling pathway have been observed. Interestingly, these mutations were observed to coexist in patterns ranging from 2-3 mutated genes to 2-4 mutations in the same gene in one sample. Further, the ''FLT3''-ITD was more prevalent in iAMP21-ALL.<ref name=":2" /> | In a 2016 paper, it was shown that in the iAMP21-ALL exome, the mutations were more commonly transitions (for example: C>T) than transversions or indels.<ref name=":0" /><ref name=":2">{{Cite journal|last=Ryan|first=S. L.|last2=Matheson|first2=E.|last3=Grossmann|first3=V.|last4=Sinclair|first4=P.|last5=Bashton|first5=M.|last6=Schwab|first6=C.|last7=Towers|first7=W.|last8=Partington|first8=M.|last9=Elliott|first9=A.|date=09 2016|title=The role of the RAS pathway in iAMP21-ALL|url=https://pubmed.ncbi.nlm.nih.gov/27168466|journal=Leukemia|volume=30|issue=9|pages=1824–1831|doi=10.1038/leu.2016.80|issn=1476-5551|pmc=5017527|pmid=27168466}}</ref> Frequently, mutations in the RAS signaling pathway have been observed. Interestingly, these mutations were observed to coexist in patterns ranging from 2-3 mutated genes to 2-4 mutations in the same gene in one sample. Further, the ''FLT3''-ITD was more prevalent in iAMP21-ALL.<ref name=":2" /> | ||
{| class="wikitable sortable" | |||
|- | |||
!Gene!!Genetic Alteration!!Tumor Suppressor Gene, Oncogene, Other!!Prevalence - | |||
Common >20%, Recurrent 5-20% or Rare <5% (Disease) | |||
!Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | |||
!Established Clinical Significance Per Guidelines - Yes or No (Source) | |||
!Clinical Relevance Details/Other Notes | |||
|- | |||
|<span class="blue-text">EXAMPLE:</span>''EGFR''<br /> | |||
|<span class="blue-text">EXAMPLE:</span> Exon 18-21 activating mutations | |||
|<span class="blue-text">EXAMPLE:</span> Oncogene | |||
|<span class="blue-text">EXAMPLE:</span> Common (lung cancer) | |||
|<span class="blue-text">EXAMPLE:</span> T | |||
|<span class="blue-text">EXAMPLE:</span> Yes (NCCN) | |||
|<span class="blue-text">EXAMPLE:</span> Exons 18, 19, and 21 mutations are targetable for therapy. Exon 20 T790M variants cause resistance to first generation TKI therapy and are targetable by second and third generation TKIs (add references). | |||
|- | |||
|<span class="blue-text">EXAMPLE:</span> ''TP53''; Variable LOF mutations<br /> | |||
|<span class="blue-text">EXAMPLE:</span> Variable LOF mutations | |||
|<span class="blue-text">EXAMPLE:</span> Tumor Supressor Gene | |||
|<span class="blue-text">EXAMPLE:</span> Common (breast cancer) | |||
|<span class="blue-text">EXAMPLE:</span> P | |||
| | |||
|<span class="blue-text">EXAMPLE:</span> >90% are somatic; rare germline alterations associated with Li-Fraumeni syndrome (add reference). Denotes a poor prognosis in breast cancer. | |||
|- | |||
|<span class="blue-text">EXAMPLE:</span> ''BRAF''; Activating mutations | |||
|<span class="blue-text">EXAMPLE:</span> Activating mutations | |||
|<span class="blue-text">EXAMPLE:</span> Oncogene | |||
|<span class="blue-text">EXAMPLE:</span> Common (melanoma) | |||
|<span class="blue-text">EXAMPLE:</span> T | |||
| | |||
| | |||
|- | |||
| | |||
| | |||
| | |||
| | |||
| | |||
| | |||
| | |||
|} | |||
Note: A more extensive list of mutations can be found in cBioportal (https://www.cbioportal.org/), COSMIC (https://cancer.sanger.ac.uk/cosmic), ICGC (https://dcc.icgc.org/) and/or other databases. When applicable, gene-specific pages within the CCGA site directly link to pertinent external content. | |||
<br /> | |||
{| class="wikitable sortable" | {| class="wikitable sortable" | ||
|- | |- | ||
| Line 188: | Line 294: | ||
| | | | ||
|} | |} | ||
<br /> | |||
==Epigenomic Alterations== | ==Epigenomic Alterations== | ||
| Line 204: | Line 310: | ||
|RAS pathway<ref name=":2" /> | |RAS pathway<ref name=":2" /> | ||
|<span class="blue-text">EXAMPLE:</span> Increased cell growth and proliferation | |<span class="blue-text">EXAMPLE:</span> Increased cell growth and proliferation | ||
|} | |} | ||
==Genetic Diagnostic Testing Methods== | ==Genetic Diagnostic Testing Methods== | ||
Revision as of 19:42, 10 October 2025
Haematolymphoid Tumours (WHO Classification, 5th ed.)
Primary Author(s)*
Holli M. Drendel, PhD, FACMGG, Carolinas Pathology Group, Charlotte
WHO Classification of Disease
| Structure | Disease |
|---|---|
| Book | Haematolymphoid Tumours (5th ed.) |
| Category | B-cell lymphoid proliferations and lymphomas |
| Family | Precursor B-cell neoplasms |
| Type | B-lymphoblastic leukaemias/lymphomas |
| Subtype(s) | B-lymphoblastic leukaemia/lymphoma with iAMP21 |
Related Terminology
| Acceptable | N/A |
| Not Recommended | N/A |
Gene Rearrangements
Some rearrangements have been seen as secondary abnormalities.
| Driver Gene | Fusion(s) and Common Partner Genes | Molecular Pathogenesis | Typical Chromosomal Alteration(s) | Prevalence -Common >20%, Recurrent 5-20% or Rare <5% (Disease) | Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | Established Clinical Significance Per Guidelines - Yes or No (Source) | Clinical Relevance Details/Other Notes |
|---|---|---|---|---|---|---|---|
| CRLF2 | P2RY8::CRLF2 | del(X)(p22.33p22.33)/del(Y)(p11.32p11.32) | Because of the unique nature of the iAMP21 abnormality, cases that present with additional genomic lesions that may suggest another category, such as a CRLF2 rearrangement, should still be classified as B-ALL with iAMP21. | ||||
| Chimeric transcription factor/fusion gene | ETV6::RUNX1 | The driver gene is a result of the fusion of 5'ETV6 and 3'RUNX1, genes that regulate hematopoiesis, which creates a chimeric transcription factor. | t(12;21)(p13.2;q22.1) | ||||
| ABL | BCR::ABL1 | The pathogenic derivative is the der(22) resulting in fusion of 5’ BCR and 3’ABL1. | t(9;22)(q34;q11.2) |
Individual Region Genomic Gain/Loss/LOH
| Chr # | Gain, Loss, Amp, LOH | Minimal Region Cytoband and/or Genomic Coordinates [Genome Build; Size] | Relevant Gene(s) | Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | Established Clinical Significance Per Guidelines - Yes or No (Source) | Clinical Relevance Details/Other Notes |
|---|---|---|---|---|---|---|
| EXAMPLE:7 | EXAMPLE: Loss | EXAMPLE:chr7 | EXAMPLE:Unknown | EXAMPLE: D, P | EXAMPLE: No | EXAMPLE:Presence of monosomy 7 (or 7q deletion) is sufficient for a diagnosis of AML with MDS-related changes when there is ≥20% blasts and no prior therapy (add reference). Monosomy 7/7q deletion is associated with a poor prognosis in AML (add references). |
| EXAMPLE:8 | EXAMPLE: Gain | EXAMPLE:chr8 | EXAMPLE:Unknown | EXAMPLE: D, P | EXAMPLE:Common recurrent secondary finding for t(8;21) (add references). | |
| EXAMPLE:17 | EXAMPLE: Amp | EXAMPLE:17q12; chr17:39,700,064-39,728,658 [hg38; 28.6 kb] | EXAMPLE:ERBB2 | EXAMPLE: D, P, T | EXAMPLE:Amplification of ERBB2 is associated with HER2 overexpression in HER2 positive breast cancer (add references). Add criteria for how amplification is defined. | |
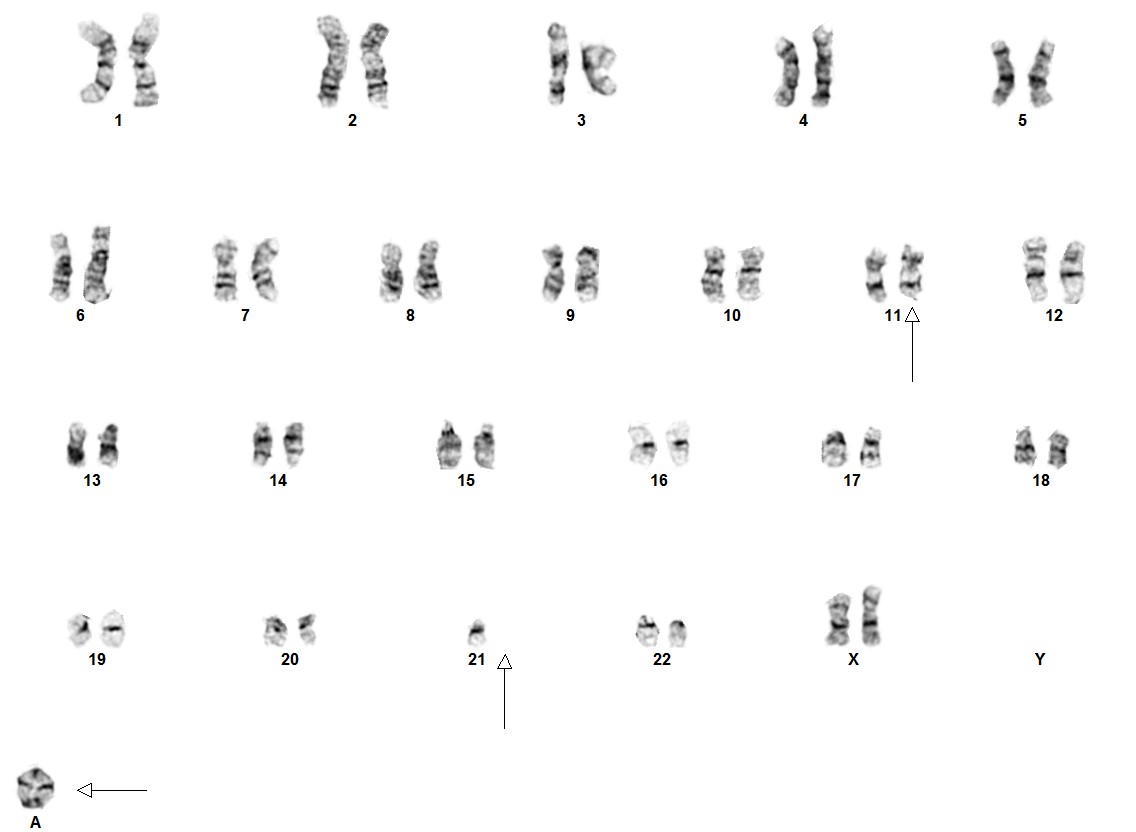
Cytogenetic morphology of the abnormal chromosome 21 can vary markedly between patients.[1] In ~80% of iAMP21 B-ALL cases, recurrent secondary abnormalities, both chromosomal and molecular, have been documented. Deletions involving particular genes such as; IKZF1, CDKN2A/B, PAX5, SH2B3, ETV6 and RB1 have also been observed.
Characteristic Chromosomal or Other Global Mutational Patterns
Put your text here and fill in the table (Instructions: Included in this category are alterations such as hyperdiploid; gain of odd number chromosomes including typically chromosome 1, 3, 5, 7, 11, and 17; co-deletion of 1p and 19q; complex karyotypes without characteristic genetic findings; chromothripsis; microsatellite instability; homologous recombination deficiency; mutational signature pattern; etc. Details on clinical significance such as prognosis and other important information can be provided in the notes section. Please include references throughout the table. Do not delete the table.)
iAMP21 cases have a characteristic pattern that is both complex and variable. This pattern comprises multiple regions of gain, amplification and deletion. Interestingly, RUNX1 amplification is not always intrachromosomal.[2][3] The formation of iAMP21 is considered to be due to breakage-fusion-bridge cycles followed by chromothripsis and other complex structural rearrangements of chromosome 21. Studies, molecular and cytogenetic, have elucidated a common 5.1 Mb region that includes the RUNX1 gene. However, even though RUNX1 is included in the amplified region, there has not yet been any conclusive evidence that RUNX1 is critical in the pathogenesis of disease given that it is not overexpressed in some individuals with this abnormality.[4][5][6]
| Chromosomal Pattern | Molecular Pathogenesis | Prevalence -
Common >20%, Recurrent 5-20% or Rare <5% (Disease) |
Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | Established Clinical Significance Per Guidelines - Yes or No (Source) | Clinical Relevance Details/Other Notes |
|---|---|---|---|---|---|
| EXAMPLE:Co-deletion of 1p and 18q | EXAMPLE: See chromosomal rearrangements table as this pattern is due to an unbalanced derivative translocation associated with oligodendroglioma (add reference). | EXAMPLE: Common (Oligodendroglioma) | EXAMPLE: D, P | ||
| EXAMPLE:Microsatellite instability - hypermutated | EXAMPLE: Common (Endometrial carcinoma) | EXAMPLE: P, T | |||
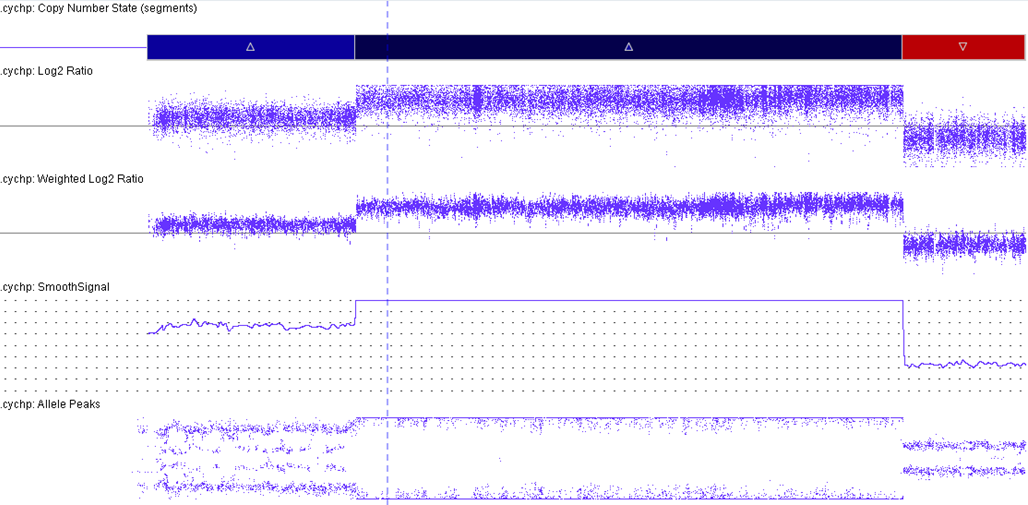
| Amplification of RUNX1 | D | See CMA image displaying the amplification of RUNX1. This is part of the critical region consistently amplified: chr21:32.8-37.9 Mb (hg19). | |||
| Terminal deletion of 21q | |||||
| Gain of X chromosome | |||||
| Abnormalities of chromosomes 7 and 11 | |||||
| Deletions of RB1 and ETV6 |
Gene Mutations (SNV/INDEL)
Put your text here and fill in the table (Instructions: This table is not meant to be an exhaustive list; please include only genes/alterations that are recurrent or common as well either disease defining and/or clinically significant. If a gene has multiple mechanisms depending on the type or site of the alteration, add multiple entries in the table. For clinical significance, denote associations with FDA-approved therapy (not an extensive list of applicable drugs) and NCCN or other national guidelines if applicable; Can also refer to CGC workgroup tables as linked on the homepage if applicable as well as any high impact papers or reviews of gene mutations in this entity. Details on clinical significance such as prognosis and other important information such as concomitant and mutually exclusive mutations can be provided in the notes section. Please include references throughout the table. Do not delete the table.)
In a 2016 paper, it was shown that in the iAMP21-ALL exome, the mutations were more commonly transitions (for example: C>T) than transversions or indels.[4][7] Frequently, mutations in the RAS signaling pathway have been observed. Interestingly, these mutations were observed to coexist in patterns ranging from 2-3 mutated genes to 2-4 mutations in the same gene in one sample. Further, the FLT3-ITD was more prevalent in iAMP21-ALL.[7]
| Gene | Genetic Alteration | Tumor Suppressor Gene, Oncogene, Other | Prevalence -
Common >20%, Recurrent 5-20% or Rare <5% (Disease) |
Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | Established Clinical Significance Per Guidelines - Yes or No (Source) | Clinical Relevance Details/Other Notes |
|---|---|---|---|---|---|---|
| EXAMPLE:EGFR |
EXAMPLE: Exon 18-21 activating mutations | EXAMPLE: Oncogene | EXAMPLE: Common (lung cancer) | EXAMPLE: T | EXAMPLE: Yes (NCCN) | EXAMPLE: Exons 18, 19, and 21 mutations are targetable for therapy. Exon 20 T790M variants cause resistance to first generation TKI therapy and are targetable by second and third generation TKIs (add references). |
| EXAMPLE: TP53; Variable LOF mutations |
EXAMPLE: Variable LOF mutations | EXAMPLE: Tumor Supressor Gene | EXAMPLE: Common (breast cancer) | EXAMPLE: P | EXAMPLE: >90% are somatic; rare germline alterations associated with Li-Fraumeni syndrome (add reference). Denotes a poor prognosis in breast cancer. | |
| EXAMPLE: BRAF; Activating mutations | EXAMPLE: Activating mutations | EXAMPLE: Oncogene | EXAMPLE: Common (melanoma) | EXAMPLE: T | ||
Note: A more extensive list of mutations can be found in cBioportal (https://www.cbioportal.org/), COSMIC (https://cancer.sanger.ac.uk/cosmic), ICGC (https://dcc.icgc.org/) and/or other databases. When applicable, gene-specific pages within the CCGA site directly link to pertinent external content.
| Gene; Genetic Alteration | Presumed Mechanism (Tumor Suppressor Gene [TSG] / Oncogene / Other) | Prevalence (COSMIC / TCGA / Other) | Concomitant Mutations | Mutually Exclusive Mutations | Diagnostic Significance (Yes, No or Unknown) | Prognostic Significance (Yes, No or Unknown) | Therapeutic Significance (Yes, No or Unknown) | Notes |
|---|---|---|---|---|---|---|---|---|
| NRAS | EXAMPLE: TSG | 45% | EXAMPLE: IDH1 R123H | EXAMPLE: EGFR amplification | EXAMPLE: Excludes hairy cell leukemia (HCL) (add reference). | |||
| KRAS | 18% | |||||||
| FLT3 | 20% | |||||||
| PTPN11 | 11% | |||||||
| BRAF | 2% | |||||||
| NF1 | 2% |
Epigenomic Alterations
N/A
Genes and Main Pathways Involved
Put your text here and fill in the table
| Gene; Genetic Alteration | Pathway | Pathophysiologic Outcome |
|---|---|---|
| RUNX1 | RAS pathway[7] | EXAMPLE: Increased cell growth and proliferation |
Genetic Diagnostic Testing Methods
Different methodologies are able to detect the iAMP21, such as:
- Fluorescence in situ hybridization (FISH) utilizing the probe set for the t(12;21)
- Conventional chromosome analysis
- Multiplex ligation-dependent probe amplification (MLPA)
- Chromosomal microarray (CMA)
- Next generation sequencing (NGS).
Familial Forms
N/A
Additional Information
This disease is defined/characterized as detailed below:
- Intrachromosomal amplification of chromosome 21 (iAMP21) is a neoplasm of lymphoblasts that are of the B-cell lineage. It is characterized by amplification of the RUNX1 gene at 21q22.3 on a structurally abnormal chromosome 21. Amplification is defined as ≥5 copies of RUNX1 detected by FISH or ≥3 copies of RUNX1 on a single abnormal chromosome 21.[8]
The epidemiology/prevalence of this disease is detailed below:
- iAMP21 is observed most often in the older pediatric group (median age of 9 years, with a range of 2-30 years). It accounts for ~2% of B-ALL cases including ~2% of standard-risk and 3% of high-risk patients. The incidence in adult B-ALL has not been established; however, it appears to be less prevalent than in the pediatric population.[4]
- Further, patients carrying a rob(15;21)(q10;q10) have an ~2700-fold increased risk of developing iAMP21 ALL compared to the general population. Additionally, patients with a constitutional ring chromosome 21, r(21), may potentially be predisposed to iAMP21 ALL.[4]
The clinical features of this disease are detailed below:
- ~50% of cases are classified as high-risk based on an age of ≥10 years.[9] Pediatric iAMP21 has been associated with a poor outcome. It displays an increased rate of relapse when treated on standard protocols. Further, the event-free survival and overall survival were significantly worse for individuals with the iAMP21 and standard-risk B-ALL, but not significant in individuals with iAMP21 and high-risk B-ALL.
- Laboratory findings - Low platelet count; Low WBC count (<50,000/μl)
The sites of involvement of this disease are detailed below:
- Bone Marrow and peripheral blood
The morphologic features of this disease are detailed below:
- There are no unique morphological or cytochemical features that distinguish this entity from other types of ALL.[8]
The immunophenotype of this disease is detailed below:
- No detailed information is known, other than these cases occur exclusively in B-ALL.[8]
Links
References
(use the "Cite" icon at the top of the page) (Instructions: Add each reference into the text above by clicking where you want to insert the reference, selecting the “Cite” icon at the top of the wiki page, and using the “Automatic” tab option to search by PMID to select the reference to insert. If a PMID is not available, such as for a book, please use the “Cite” icon, select “Manual” and then “Basic Form”, and include the entire reference. To insert the same reference again later in the page, select the “Cite” icon and “Re-use” to find the reference; DO NOT insert the same reference twice using the “Automatic” tab as it will be treated as two separate references. The reference list in this section will be automatically generated and sorted.)
- ↑ Harewood, L.; et al. (2003-03). "Amplification of AML1 on a duplicated chromosome 21 in acute lymphoblastic leukemia: a study of 20 cases". Leukemia. 17 (3): 547–553. doi:10.1038/sj.leu.2402849. ISSN 0887-6924. PMID 12646943. Check date values in:
|date=(help) - ↑ Arber, Daniel A. (04 2019). "The 2016 WHO classification of acute myeloid leukemia: What the practicing clinician needs to know". Seminars in Hematology. 56 (2): 90–95. doi:10.1053/j.seminhematol.2018.08.002. ISSN 1532-8686. PMID 30926096. Check date values in:
|date=(help) - ↑ Johnson, Ryan C.; et al. (2015-07). "Cytogenetic Variation of B-Lymphoblastic Leukemia With Intrachromosomal Amplification of Chromosome 21 (iAMP21): A Multi-Institutional Series Review". American Journal of Clinical Pathology. 144 (1): 103–112. doi:10.1309/AJCPLUYF11HQBYRB. ISSN 1943-7722. PMID 26071468. Check date values in:
|date=(help) - ↑ 4.0 4.1 4.2 4.3 Akkari, Yassmine M. N.; et al. (2020-05). "Evidence-based review of genomic aberrations in B-lymphoblastic leukemia/lymphoma: Report from the cancer genomics consortium working group for lymphoblastic leukemia". Cancer Genetics. 243: 52–72. doi:10.1016/j.cancergen.2020.03.001. ISSN 2210-7762. PMID 32302940 Check
|pmid=value (help). Check date values in:|date=(help) - ↑ Rand, Vikki; et al. (2011-06-23). "Genomic characterization implicates iAMP21 as a likely primary genetic event in childhood B-cell precursor acute lymphoblastic leukemia". Blood. 117 (25): 6848–6855. doi:10.1182/blood-2011-01-329961. ISSN 1528-0020. PMID 21527530.
- ↑ Hunger, Stephen P.; et al. (2012-05-10). "Improved survival for children and adolescents with acute lymphoblastic leukemia between 1990 and 2005: a report from the children's oncology group". Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology. 30 (14): 1663–1669. doi:10.1200/JCO.2011.37.8018. ISSN 1527-7755. PMC 3383113. PMID 22412151.
- ↑ 7.0 7.1 7.2 Ryan, S. L.; et al. (09 2016). "The role of the RAS pathway in iAMP21-ALL". Leukemia. 30 (9): 1824–1831. doi:10.1038/leu.2016.80. ISSN 1476-5551. PMC 5017527. PMID 27168466. Check date values in:
|date=(help) - ↑ 8.0 8.1 8.2 Borowitz MJ, et al., (2017). B-Lymphoblastic leukaemia/lymphoma with recurrent genetic abnormalities, in World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised 4th edition. Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Arber DA, Hasserjian RP, Le Beau MM, Orazi A, and Siebert R, Editors. IARC Press: Lyon, France.
- ↑ Harrison, Christine J. (2015-02-26). "Blood Spotlight on iAMP21 acute lymphoblastic leukemia (ALL), a high-risk pediatric disease". Blood. 125 (9): 1383–1386. doi:10.1182/blood-2014-08-569228. ISSN 1528-0020. PMID 25608562.
Notes
*Primary authors will typically be those that initially create and complete the content of a page. If a subsequent user modifies the content and feels the effort put forth is of high enough significance to warrant listing in the authorship section, please contact the Associate Editor or other CCGA representative. When pages have a major update, the new author will be acknowledged at the beginning of the page, and those who contributed previously will be acknowledged below as a prior author.
Prior Author(s):
*Citation of this Page: “B-lymphoblastic leukaemia/lymphoma with iAMP21”. Compendium of Cancer Genome Aberrations (CCGA), Cancer Genomics Consortium (CGC), updated 10/10/2025, https://ccga.io/index.php/HAEM5:B-lymphoblastic_leukaemia/lymphoma_with_iAMP21.