HAEM5:Acute myeloid leukaemia with NUP98 rearrangement: Difference between revisions
| [pending revision] | [pending revision] |
mNo edit summary |
mNo edit summary |
||
| Line 46: | Line 46: | ||
==Gene Rearrangements== | ==Gene Rearrangements== | ||
Acute myeloid leukemia (AML) with ''NUP98'' rearrangement is characterized by chromosomal translocations involving ''NUP98'' (nucleoporin 98 and 96 precursor) on chromosome 11p15.4 and various partner genes - more than 40 of such have been reported to date.<ref>Patkar N, Meshinchi S, Westerman D, et al. Acute myeloid leukaemia with NUP98 rearrangement. In: WHO Classification of Tumours Editorial Board. Haematolymphoid tumours. Lyon (France): International Agency for Research on Cancer; 2024. . (WHO classification of tumours series, 5th ed.; vol. 11). <nowiki>https://publications.iarc.who.int/637</nowiki>.</ref> The ''NUP98'' gene encodes protein component of the nuclear pore complex which facilitates nucleocytoplasmic transport of RNA and has roles in transcriptional and cell cycle regulation.<ref name=":2">{{Cite journal|last=Gough|first=Sheryl M.|last2=Slape|first2=Christopher I.|last3=Aplan|first3=Peter D.|date=2011-12-08|title=NUP98 gene fusions and hematopoietic malignancies: common themes and new biologic insights|url=https://pubmed.ncbi.nlm.nih.gov/21948299|journal=Blood|volume=118|issue=24|pages=6247–6257|doi=10.1182/blood-2011-07-328880|issn=1528-0020|pmc=3236115|pmid=21948299}}</ref><ref>{{Cite journal|last=Michmerhuizen|first=Nicole L.|last2=Klco|first2=Jeffery M.|last3=Mullighan|first3=Charles G.|date=2020-11-12|title=Mechanistic insights and potential therapeutic approaches for NUP98-rearranged hematologic malignancies|url=https://pubmed.ncbi.nlm.nih.gov/32766874|journal=Blood|volume=136|issue=20|pages=2275–2289|doi=10.1182/blood.2020007093|issn=1528-0020|pmc=7702474|pmid=32766874}}</ref> NUP98 fusion proteins typically involve the N-terminal portion of NUP98 and the C-terminal portion of the fusion partner.<ref name=":3">{{Cite journal|last=Gough|first=Sheryl M.|last2=Slape|first2=Christopher I.|last3=Aplan|first3=Peter D.|date=2011-12-08|title=NUP98 gene fusions and hematopoietic malignancies: common themes and new biologic insights|url=https://pubmed.ncbi.nlm.nih.gov/21948299|journal=Blood|volume=118|issue=24|pages=6247–6257|doi=10.1182/blood-2011-07-328880|issn=1528-0020|pmc=3236115|pmid=21948299}}</ref> Fusion partners commonly include transcription factors (such as ''HOX'' elements, most often ''HOXA9'') or epigenetic regulators (most commonly involving ''NSD1'' or ''KDM5A''), however a range of partners belonging to neither of these categories has been identified, many of which contain coiled-coil domains thought to facilitate oligomerization.<ref name=":0">{{Cite journal|last=Mohanty|first=Sagarajit|date=2023-09|title=NUP98 Rearrangements in AML: Molecular Mechanisms and Clinical Implications|url=https://www.mdpi.com/2673-7523/3/3/11|journal=Onco|language=en|volume=3|issue=3|pages=147–164|doi=10.3390/onco3030011|issn=2673-7523}}</ref><ref name=":3" /> | |||
Acute myeloid leukemia (AML) with ''NUP98'' rearrangement is characterized by chromosomal translocations involving ''NUP98'' (nucleoporin 98 and 96 precursor) on chromosome 11p15.4 and various partner genes - more than 40 of such have been reported to date.<ref>Patkar N, Meshinchi S, Westerman D, et al. Acute myeloid leukaemia with NUP98 rearrangement. In: WHO Classification of Tumours Editorial Board. Haematolymphoid tumours. Lyon (France): International Agency for Research on Cancer; 2024. . (WHO classification of tumours series, 5th ed.; vol. 11). <nowiki>https://publications.iarc.who.int/637</nowiki>.</ref> The ''NUP98'' gene encodes protein component of the nuclear pore complex which facilitates nucleocytoplasmic transport of | |||
< | |||
{| class="wikitable" | {| class="wikitable" | ||
|'''Driver Gene''' | |'''Driver Gene''' | ||
| Line 123: | Line 59: | ||
|''NUP98'' | |''NUP98'' | ||
|''NUP98::NSD1'' | |''NUP98::NSD1'' | ||
|Fusion of N-terminal ''NUP98'' (with fusion junction most often involving exons 12-13) to C-terminal ''NSD1''; fusion proteins result in epigenetic modification and dysregulation of ''HOXA''/''HOXB'' family genes, among others, through functional domains in both fusion partners. | |||
|NUP98-NSD1 | |||
|t(5;11)(q35;p15) | |t(5;11)(q35;p15) | ||
| Line 130: | Line 65: | ||
|Rare (AML) | |Rare (AML) | ||
|Defining genetic abnormality in AML | |Defining genetic abnormality in AML | ||
|Yes (WHO) | |Yes (WHO/ICC) | ||
|Rare | |Rare though most common recurrent NUP98 rearrangement in children and young adults.<ref name=":1">{{Cite journal|last=Bertrums|first=Eline J. M.|last2=Smith|first2=Jenny L.|last3=Harmon|first3=Lauren|last4=Ries|first4=Rhonda E.|last5=Wang|first5=Yi-Cheng J.|last6=Alonzo|first6=Todd A.|last7=Menssen|first7=Andrew J.|last8=Chisholm|first8=Karen M.|last9=Leonti|first9=Amanda R.|date=2023-02-23|title=Comprehensive molecular and clinical characterization of NUP98 fusions in pediatric acute myeloid leukemia|url=https://www.haematologica.org/article/view/haematol.2022.281653|journal=Haematologica|language=en|volume=108|issue=8|pages=2044–2058|doi=10.3324/haematol.2022.281653|issn=1592-8721}}</ref> | ||
|- | |- | ||
|''NUP98'' | |''NUP98'' | ||
|''NUP98::KDM5A'' | |''NUP98::KDM5A'' | ||
|KDM5A | |Fusion of N-terminal ''NUP98'' (fusion junction most often involving exons 13-14) to C-terminal ''KDM5A;'' fusion proteins result in epigenetic modification and dysregulation of ''HOXA''/''HOXB'' family genes, among others, through functional domains in both fusion partners. | ||
|t(11;12)(p15;p13) | |t(11;12)(p15;p13) | ||
| Line 141: | Line 76: | ||
|Rare (AML) | |Rare (AML) | ||
|Defining genetic abnormality in AML | |Defining genetic abnormality in AML | ||
|Yes (WHO) | |Yes (WHO/ICC) | ||
| | |Commonly associated with erythroid and megakaryocytic phenotypes in pediatric AML (acute erythroid leukemia and acute megakaryocytic leukemia). <ref name=":1" /> | ||
|- | |- | ||
|''NUP98'' | |''NUP98'' | ||
|''NUP98::HOXA9'' | |''NUP98::HOXA9'' | ||
|Fusion of N-terminal ''NUP98'' (fusion junction most often involving exons 13-14) to C-terminal ''HOXA9;'' fusion proteins result in epigenetic modification and dysregulation of ''HOXA''/''HOXB'' family genes, among others, through functional domains in both fusion partners. | |||
|NUP98 | |t(7;11)(p15;p15) | ||
|t(7;11)(p15 | |||
|Rare (AML) | |Rare (AML) | ||
|Defining genetic abnormality in AML | |Defining genetic abnormality in AML | ||
| | |Yes (WHO/ICC) | ||
| | | | ||
|} | |} | ||
==Individual Region Genomic Gain/Loss/LOH== | ==Individual Region Genomic Gain/Loss/LOH== | ||
{| class="wikitable" | {| class="wikitable" | ||
|''' | |'''Chr#''' | ||
|'''Gain/Loss/Amp/LOH''' | |'''Gain/Loss/Amp/LOH''' | ||
|'''Minimal Region Cytoband and/or Genomic Coordinates [Genome Build; Size]''' | |'''Minimal Region Cytoband and/or Genomic Coordinates [Genome Build; Size]''' | ||
| Line 233: | Line 108: | ||
|- | |- | ||
|13 | |13 | ||
| | |Loss | ||
| | |13q14.2q14.3<ref name=":2" /> | ||
|RB1 | |''RB1'' | ||
|NA | |NA | ||
| | |No | ||
| | |Highly enriched in ''NUP98::KDM5A'' | ||
|} | |} | ||
| Line 322: | Line 197: | ||
| | | | ||
| | | | ||
|} | |}FLT3-ITD and WT1 mutation are recurring events in NUP98::NSD1 and was also observed in some NUP98::HOXA9 AML patients.(R1). Loss of RB1 at 13q14 is particularly associated with NUP98::KDM5A | ||
FLT3-ITD and WT1 mutation are recurring events in NUP98::NSD1 and was also observed in some NUP98::HOXA9 AML patients.(R1). Loss of RB1 at 13q14 is particularly associated with NUP98::KDM5A | |||
{| class="wikitable" | {| class="wikitable" | ||
|'''Gene''' | |'''Gene''' | ||
| Line 334: | Line 207: | ||
|'''Clinical Relevance Details/Other Notes''' | |'''Clinical Relevance Details/Other Notes''' | ||
|- | |- | ||
|''FLT3 | |''FLT3'' | ||
| | |Internal tandem duplication | ||
| | |Oncogene | ||
| | |Common (frequency varies with fusion partner) | ||
| | |P,T | ||
| | | | ||
|Seen in 67 to 91% of cases with NUP98::NSD1 | |Seen in 67 to 91% of cases with NUP98::NSD1 | ||
|- | |- | ||
|''WT1'' | |''WT1'' | ||
|Gain or loss of function | |||
|Oncogene/Tumor Suppressor Gene | |||
| | | | ||
| | | | ||
| | | | ||
|Reported in 33-55% of NUP98::NSD1 rearranged AML | |Reported in 33-55% of NUP98::NSD1 rearranged AML | ||
|} | |} | ||
Note: A more extensive list of mutations can be found in [https://www.cbioportal.org/ <u>cBioportal</u>], [https://cancer.sanger.ac.uk/cosmic <u>COSMIC</u>], and/or other databases. When applicable, gene-specific pages within the CCGA site directly link to pertinent external content. | |||
==Epigenomic Alterations== | ==Epigenomic Alterations== | ||
Revision as of 15:28, 30 December 2025
Haematolymphoid Tumours (WHO Classification, 5th ed.)
| This page is under construction |
(General Instructions – The focus of these pages is the clinically significant genetic alterations in each disease type. This is based on up-to-date knowledge from multiple resources such as PubMed and the WHO classification books. The CCGA is meant to be a supplemental resource to the WHO classification books; the CCGA captures in a continually updated wiki-stye manner the current genetics/genomics knowledge of each disease, which evolves more rapidly than books can be revised and published. If the same disease is described in multiple WHO classification books, the genetics-related information for that disease will be consolidated into a single main page that has this template (other pages would only contain a link to this main page). Use HUGO-approved gene names and symbols (italicized when appropriate), HGVS-based nomenclature for variants, as well as generic names of drugs and testing platforms or assays if applicable. Please complete tables whenever possible and do not delete them (add N/A if not applicable in the table and delete the examples); to add (or move) a row or column in a table, click nearby within the table and select the > symbol that appears. Please do not delete or alter the section headings. The use of bullet points alongside short blocks of text rather than only large paragraphs is encouraged. Additional instructions below in italicized blue text should not be included in the final page content. Please also see Author_Instructions and FAQs as well as contact your Associate Editor or Technical Support.)
Primary Author(s)*
Eric McGinnis, MD
Fatma Albulushi, MD
WHO Classification of Disease
| Structure | Disease |
|---|---|
| Book | Haematolymphoid Tumours (5th ed.) |
| Category | Myeloid proliferations and neoplasms |
| Family | Acute myeloid leukaemia |
| Type | Acute myeloid leukaemia with defining genetic abnormalities |
| Subtype(s) | Acute myeloid leukaemia with NUP98 rearrangement |
Related Terminology
| Acceptable | N/A |
| Not Recommended | N/A |
Gene Rearrangements
Acute myeloid leukemia (AML) with NUP98 rearrangement is characterized by chromosomal translocations involving NUP98 (nucleoporin 98 and 96 precursor) on chromosome 11p15.4 and various partner genes - more than 40 of such have been reported to date.[1] The NUP98 gene encodes protein component of the nuclear pore complex which facilitates nucleocytoplasmic transport of RNA and has roles in transcriptional and cell cycle regulation.[2][3] NUP98 fusion proteins typically involve the N-terminal portion of NUP98 and the C-terminal portion of the fusion partner.[4] Fusion partners commonly include transcription factors (such as HOX elements, most often HOXA9) or epigenetic regulators (most commonly involving NSD1 or KDM5A), however a range of partners belonging to neither of these categories has been identified, many of which contain coiled-coil domains thought to facilitate oligomerization.[5][4]
| Driver Gene | Fusion(s) and Common Partner Genes | Molecular Pathogenesis | Typical Chromosomal Alteration(s) | Prevalence -Common >20%, Recurrent 5-20% or Rare <5% (Disease) | Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | Established Clinical Significance Per Guidelines - Yes or No (Source) | Clinical Relevance Details/Other Notes |
| NUP98 | NUP98::NSD1 | Fusion of N-terminal NUP98 (with fusion junction most often involving exons 12-13) to C-terminal NSD1; fusion proteins result in epigenetic modification and dysregulation of HOXA/HOXB family genes, among others, through functional domains in both fusion partners. | t(5;11)(q35;p15)
Usually cryptic |
Rare (AML) | Defining genetic abnormality in AML | Yes (WHO/ICC) | Rare though most common recurrent NUP98 rearrangement in children and young adults.[6] |
| NUP98 | NUP98::KDM5A | Fusion of N-terminal NUP98 (fusion junction most often involving exons 13-14) to C-terminal KDM5A; fusion proteins result in epigenetic modification and dysregulation of HOXA/HOXB family genes, among others, through functional domains in both fusion partners. | t(11;12)(p15;p13)
Usually cryptic |
Rare (AML) | Defining genetic abnormality in AML | Yes (WHO/ICC) | Commonly associated with erythroid and megakaryocytic phenotypes in pediatric AML (acute erythroid leukemia and acute megakaryocytic leukemia). [6] |
| NUP98 | NUP98::HOXA9 | Fusion of N-terminal NUP98 (fusion junction most often involving exons 13-14) to C-terminal HOXA9; fusion proteins result in epigenetic modification and dysregulation of HOXA/HOXB family genes, among others, through functional domains in both fusion partners. | t(7;11)(p15;p15) | Rare (AML) | Defining genetic abnormality in AML | Yes (WHO/ICC) |
Individual Region Genomic Gain/Loss/LOH
| Chr# | Gain/Loss/Amp/LOH | Minimal Region Cytoband and/or Genomic Coordinates [Genome Build; Size] | Relevant Gene(s) | Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | Established Clinical Significance Per Guidelines - Yes or No (Source) | Clinical Relevance Details/Other Notes |
| 8 | Gain | Trisomy 8 | Unknown | NA | No | |
| 13 | Loss | 13q14.2q14.3[2] | RB1 | NA | No | Highly enriched in NUP98::KDM5A |
Characteristic Chromosomal or Other Global Mutational Patterns
Put your text here and fill in the table (Instructions: Included in this category are alterations such as hyperdiploid; gain of odd number chromosomes including typically chromosome 1, 3, 5, 7, 11, and 17; co-deletion of 1p and 19q; complex karyotypes without characteristic genetic findings; chromothripsis; microsatellite instability; homologous recombination deficiency; mutational signature pattern; etc. Details on clinical significance such as prognosis and other important information can be provided in the notes section. Please include references throughout the table. Do not delete the table.)
| Chromosomal Pattern | Molecular Pathogenesis | Prevalence -
Common >20%, Recurrent 5-20% or Rare <5% (Disease) |
Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | Established Clinical Significance Per Guidelines - Yes or No (Source) | Clinical Relevance Details/Other Notes |
|---|---|---|---|---|---|
| EXAMPLE:
Co-deletion of 1p and 18q |
EXAMPLE: See chromosomal rearrangements table as this pattern is due to an unbalanced derivative translocation associated with oligodendroglioma (add reference). | EXAMPLE: Common (Oligodendroglioma) | EXAMPLE: D, P | ||
| EXAMPLE:
Microsatellite instability - hypermutated |
EXAMPLE: Common (Endometrial carcinoma) | EXAMPLE: P, T | |||
Gene Mutations (SNV/INDEL)
Put your text here and fill in the table (Instructions: This table is not meant to be an exhaustive list; please include only genes/alterations that are recurrent or common as well either disease defining and/or clinically significant. If a gene has multiple mechanisms depending on the type or site of the alteration, add multiple entries in the table. For clinical significance, denote associations with FDA-approved therapy (not an extensive list of applicable drugs) and NCCN or other national guidelines if applicable; Can also refer to CGC workgroup tables as linked on the homepage if applicable as well as any high impact papers or reviews of gene mutations in this entity. Details on clinical significance such as prognosis and other important information such as concomitant and mutually exclusive mutations can be provided in the notes section. Please include references throughout the table. Do not delete the table.)
| Gene | Genetic Alteration | Tumor Suppressor Gene, Oncogene, Other | Prevalence -
Common >20%, Recurrent 5-20% or Rare <5% (Disease) |
Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | Established Clinical Significance Per Guidelines - Yes or No (Source) | Clinical Relevance Details/Other Notes |
|---|---|---|---|---|---|---|
| EXAMPLE:EGFR
|
EXAMPLE: Exon 18-21 activating mutations | EXAMPLE: Oncogene | EXAMPLE: Common (lung cancer) | EXAMPLE: T | EXAMPLE: Yes (NCCN) | EXAMPLE: Exons 18, 19, and 21 mutations are targetable for therapy. Exon 20 T790M variants cause resistance to first generation TKI therapy and are targetable by second and third generation TKIs (add references). |
| EXAMPLE: TP53; Variable LOF mutations
|
EXAMPLE: Variable LOF mutations | EXAMPLE: Tumor Supressor Gene | EXAMPLE: Common (breast cancer) | EXAMPLE: P | EXAMPLE: >90% are somatic; rare germline alterations associated with Li-Fraumeni syndrome (add reference). Denotes a poor prognosis in breast cancer. | |
| EXAMPLE: BRAF; Activating mutations | EXAMPLE: Activating mutations | EXAMPLE: Oncogene | EXAMPLE: Common (melanoma) | EXAMPLE: T | ||
FLT3-ITD and WT1 mutation are recurring events in NUP98::NSD1 and was also observed in some NUP98::HOXA9 AML patients.(R1). Loss of RB1 at 13q14 is particularly associated with NUP98::KDM5A
| Gene | Genetic Alteration | Tumor Suppressor Gene (TSG)/Oncogene/Other | Prevalence -Common >20%, Recurrent 5-20% or Rare <5% (Disease) | Diagnostic, Prognostic, and Therapeutic Significance - D, P, T | Established Clinical Significance Per Guidelines - Yes or No (Source) | Clinical Relevance Details/Other Notes |
| FLT3 | Internal tandem duplication | Oncogene | Common (frequency varies with fusion partner) | P,T | Seen in 67 to 91% of cases with NUP98::NSD1 | |
| WT1 | Gain or loss of function | Oncogene/Tumor Suppressor Gene | Reported in 33-55% of NUP98::NSD1 rearranged AML |
Note: A more extensive list of mutations can be found in cBioportal, COSMIC, and/or other databases. When applicable, gene-specific pages within the CCGA site directly link to pertinent external content.
Epigenomic Alterations
Put your text here
Genes and Main Pathways Involved
Put your text here and fill in the table (Instructions: Please include references throughout the table. Do not delete the table.)
| Gene; Genetic Alteration | Pathway | Pathophysiologic Outcome |
|---|---|---|
| EXAMPLE: BRAF and MAP2K1; Activating mutations | EXAMPLE: MAPK signaling | EXAMPLE: Increased cell growth and proliferation |
| EXAMPLE: CDKN2A; Inactivating mutations | EXAMPLE: Cell cycle regulation | EXAMPLE: Unregulated cell division |
| EXAMPLE: KMT2C and ARID1A; Inactivating mutations | EXAMPLE: Histone modification, chromatin remodeling | EXAMPLE: Abnormal gene expression program |
Genetic Diagnostic Testing Methods
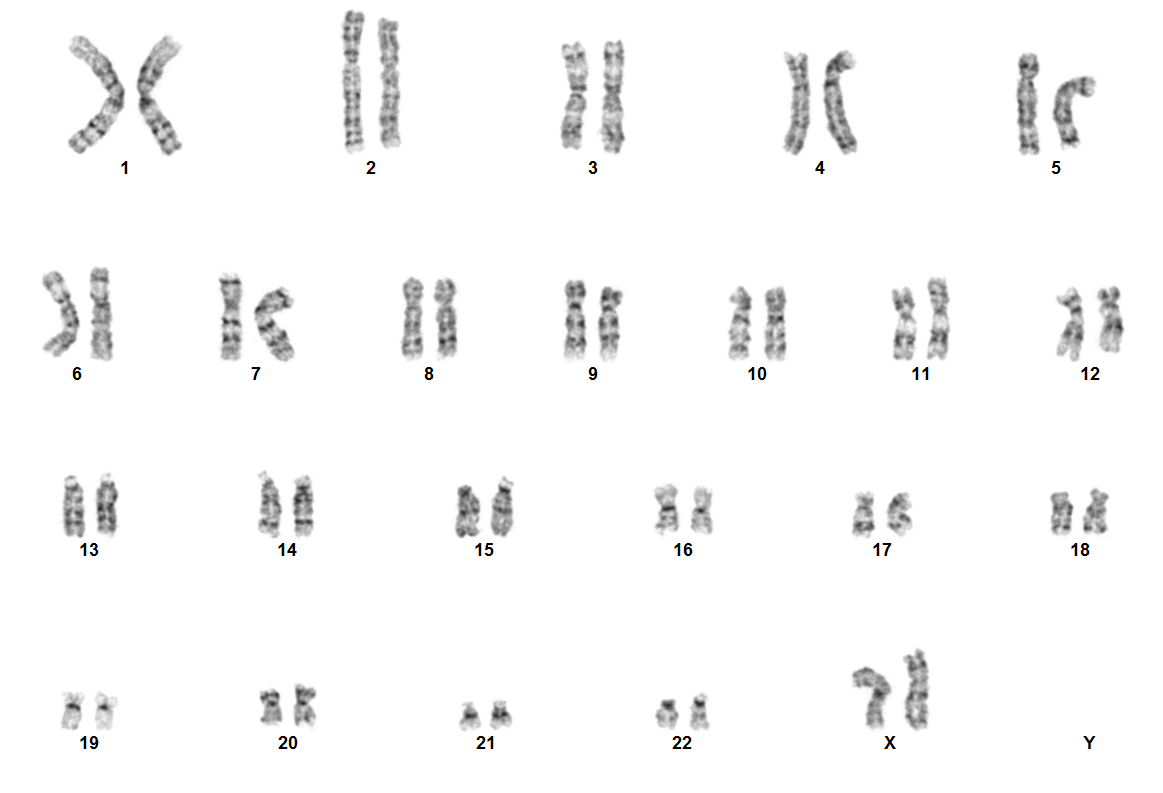
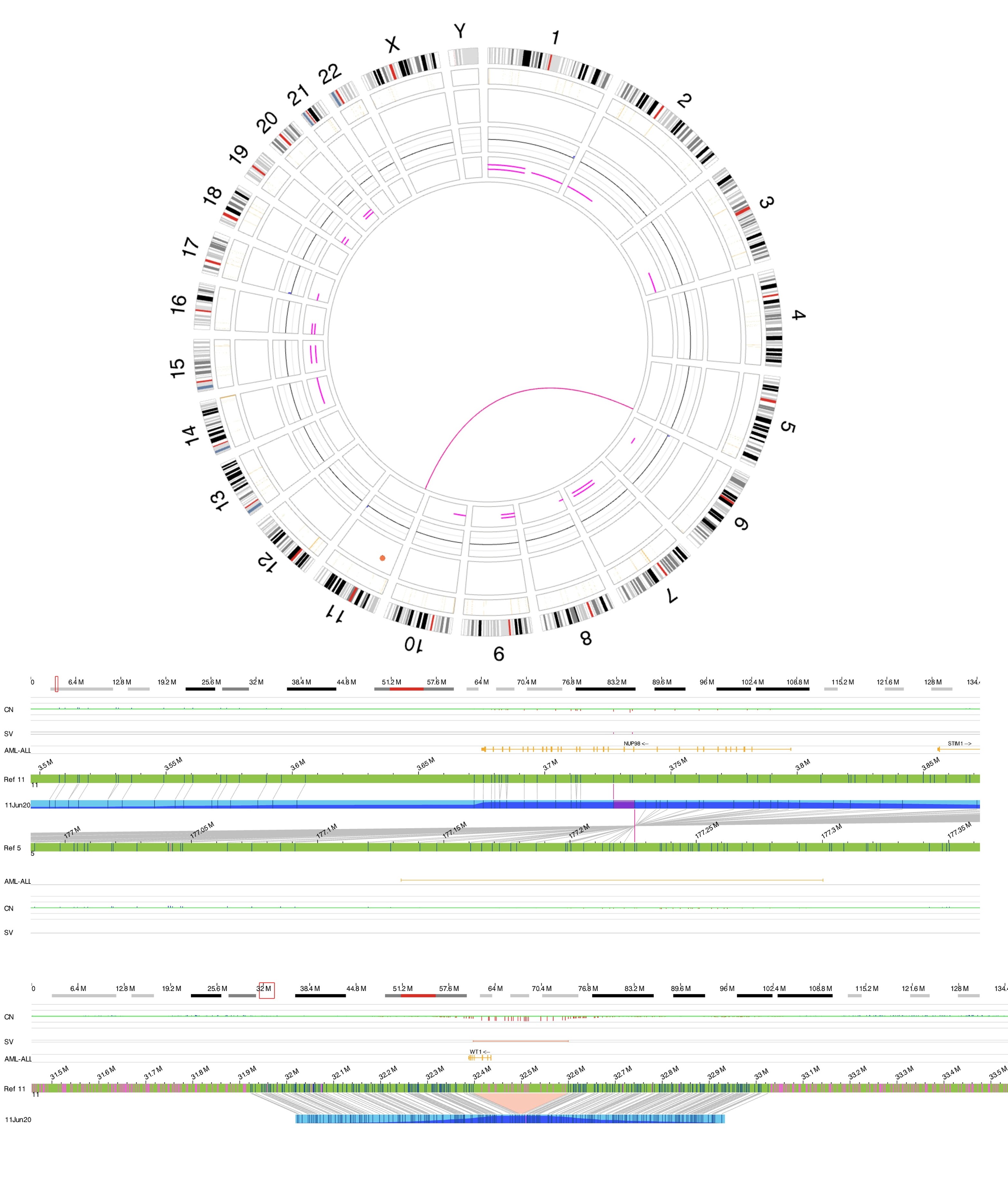
Rearrangements involving NUP98 are often cryptic on conventional karyotype, owing to terminal location of NUP98 on chromosome 11p15.4. Most patients have a normal karyotype. Diagnosis is established using the following tests:
- FISH using NUP98 break-apart probes
- RT-PCR for fusion proteins like NUP98::NSD1
- RNA sequencing
- Optical Genome Mapping (OGM)
Familial Forms
Put your text here (Instructions: Include associated hereditary conditions/syndromes that cause this entity or are caused by this entity.)
Additional Information
Links
Put a link here or anywhere appropriate in this page (Instructions: Highlight the text to which you want to add a link in this section or elsewhere, select the "Link" icon at the top of the wiki page, and search the name of the internal page to which you want to link this text, or enter an external internet address by including the "http://www." portion.)
References
(use the "Cite" icon at the top of the page) (Instructions: Add each reference into the text above by clicking where you want to insert the reference, selecting the “Cite” icon at the top of the wiki page, and using the “Automatic” tab option to search by PMID to select the reference to insert. If a PMID is not available, such as for a book, please use the “Cite” icon, select “Manual” and then “Basic Form”, and include the entire reference. To insert the same reference again later in the page, select the “Cite” icon and “Re-use” to find the reference; DO NOT insert the same reference twice using the “Automatic” tab as it will be treated as two separate references. The reference list in this section will be automatically generated and sorted.)
Notes
*Primary authors will typically be those that initially create and complete the content of a page. If a subsequent user modifies the content and feels the effort put forth is of high enough significance to warrant listing in the authorship section, please contact the Associate Editor or other CCGA representative. When pages have a major update, the new author will be acknowledged at the beginning of the page, and those who contributed previously will be acknowledged below as a prior author.
Prior Author(s):
*Citation of this Page: “Acute myeloid leukaemia with NUP98 rearrangement”. Compendium of Cancer Genome Aberrations (CCGA), Cancer Genomics Consortium (CGC), updated 12/30/2025, https://ccga.io/index.php/HAEM5:Acute_myeloid_leukaemia_with_NUP98_rearrangement.
- ↑ Patkar N, Meshinchi S, Westerman D, et al. Acute myeloid leukaemia with NUP98 rearrangement. In: WHO Classification of Tumours Editorial Board. Haematolymphoid tumours. Lyon (France): International Agency for Research on Cancer; 2024. . (WHO classification of tumours series, 5th ed.; vol. 11). https://publications.iarc.who.int/637.
- ↑ 2.0 2.1 Gough, Sheryl M.; et al. (2011-12-08). "NUP98 gene fusions and hematopoietic malignancies: common themes and new biologic insights". Blood. 118 (24): 6247–6257. doi:10.1182/blood-2011-07-328880. ISSN 1528-0020. PMC 3236115. PMID 21948299.
- ↑ Michmerhuizen, Nicole L.; et al. (2020-11-12). "Mechanistic insights and potential therapeutic approaches for NUP98-rearranged hematologic malignancies". Blood. 136 (20): 2275–2289. doi:10.1182/blood.2020007093. ISSN 1528-0020. PMC 7702474 Check
|pmc=value (help). PMID 32766874 Check|pmid=value (help). - ↑ 4.0 4.1 Gough, Sheryl M.; et al. (2011-12-08). "NUP98 gene fusions and hematopoietic malignancies: common themes and new biologic insights". Blood. 118 (24): 6247–6257. doi:10.1182/blood-2011-07-328880. ISSN 1528-0020. PMC 3236115. PMID 21948299.
- ↑ Mohanty, Sagarajit (2023-09). "NUP98 Rearrangements in AML: Molecular Mechanisms and Clinical Implications". Onco. 3 (3): 147–164. doi:10.3390/onco3030011. ISSN 2673-7523. Check date values in:
|date=(help) - ↑ 6.0 6.1 Bertrums, Eline J. M.; et al. (2023-02-23). "Comprehensive molecular and clinical characterization of NUP98 fusions in pediatric acute myeloid leukemia". Haematologica. 108 (8): 2044–2058. doi:10.3324/haematol.2022.281653. ISSN 1592-8721.